
Azathioprine and Dental Health: Essential Guide for Patients
Learn how azathioprine affects oral health, what dental risks to watch for, and practical steps to keep your mouth safe while on this immunosuppressant.
One simple blood test before you start azathioprine can prevent very serious side effects. That makes the medicine both powerful and picky. Azathioprine is an immune-suppressing drug used for autoimmune diseases (like Crohn's, rheumatoid arthritis) and to prevent organ rejection. It can calm a runaway immune system, but it needs regular lab checks and a few safety rules to use well.
Azathioprine turns into active metabolites that reduce how fast certain white blood cells grow. That lowers inflammation and the immune attack in conditions such as inflammatory bowel disease, lupus, and after kidney or liver transplant. Doctors often start it when other medicines don't control symptoms or when long-term immune control is needed.
Expect slow results — it usually takes 8–12 weeks to see clear benefit. That means azathioprine is more of a steady, long-term option rather than a fast-acting rescue drug.
Common short-term effects include nausea, tiredness, and mild stomach upset. Important but less common problems include low white blood cells (which raises infection risk), liver enzyme increases, and pancreatitis. There’s also a small increased risk of certain cancers with long-term use. Watch for fever, sore throat, easy bruising, dark urine, yellowing of the skin, or severe stomach pain — these need immediate medical attention.
Two genetic tests — TPMT and NUDT15 — are useful before starting. If you have low TPMT or NUDT15 activity, you can get severe bone marrow suppression from normal doses. Many clinics now run at least one of these tests so dosing can be safer from day one.
Azathioprine interacts notably with allopurinol (used for gout). If you take allopurinol, your azathioprine dose should be cut to about a quarter — otherwise you risk serious bone marrow problems. Avoid live vaccines while your immune system is suppressed. Tell your doctor about all medicines and supplements you take.
Monitoring schedule (typical):
- Baseline: CBC (complete blood count), liver tests, and TPMT/NUDT15 if available.
- First 1–2 months: CBC and LFTs every 1–2 weeks.
- After stability: monthly for 2–3 months, then every 3 months or as advised by your doctor.
Dosing is weight-based. Typical maintenance ranges are about 1–2 mg/kg/day for autoimmune disease; higher initiation doses are used in some transplant settings. Exact dose depends on your condition, tests, and doctor’s plan — do not self-adjust.
Pregnancy and breastfeeding: some specialists continue azathioprine in pregnancy when benefits outweigh risks. Mycophenolate is usually avoided in pregnancy because it’s more clearly linked to birth defects. Always discuss pregnancy plans with your provider.
Bottom line: azathioprine can control tough immune problems but needs pre-tests, regular labs, and attention to drug interactions. Ask about TPMT/NUDT15 testing, learn the warning signs to watch for, and keep a clear follow-up plan with your clinician.

Learn how azathioprine affects oral health, what dental risks to watch for, and practical steps to keep your mouth safe while on this immunosuppressant.
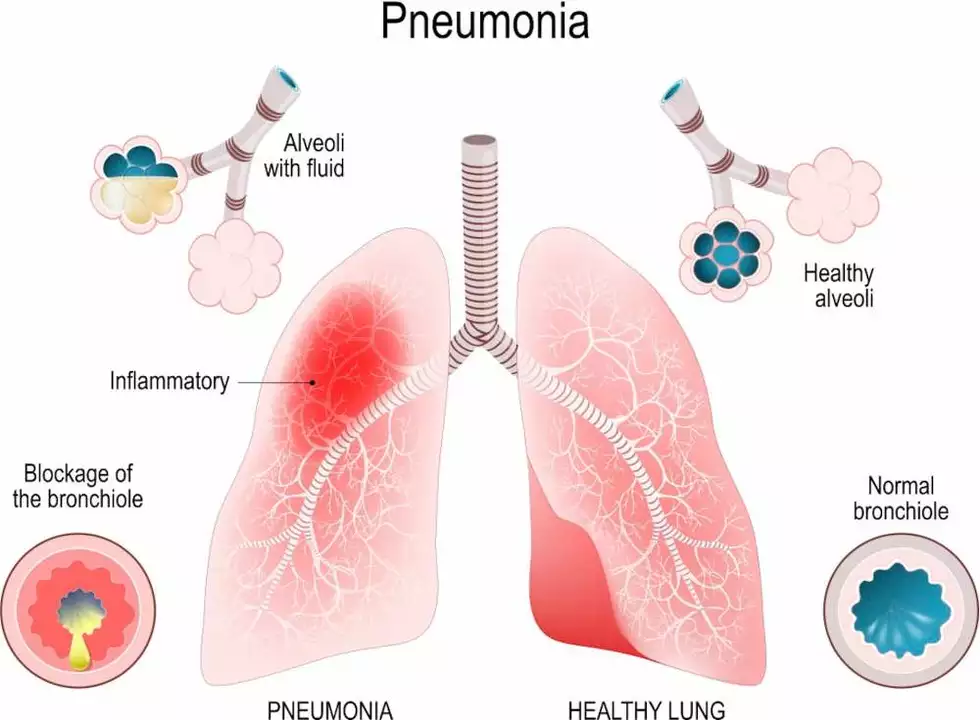
In a recent comprehensive review, I explored the connection between Azathioprine and the risk of pneumonia. Azathioprine, an immunosuppressive drug, has been linked to increased vulnerability to infections. I discovered that patients taking this medication, particularly those with autoimmune or inflammatory disorders, may have a heightened risk of developing pneumonia. It is crucial for healthcare professionals to closely monitor these patients and weigh the potential benefits and risks of using Azathioprine. Ultimately, the goal is to ensure the safest and most effective treatment plan for each individual.